Clinical trials are essential — but patients don’t live in controlled environments. They receive care in busy clinics, manage other conditions, and balance treatment with everyday life.
Real-World Data (RWD) helps bring that reality into trial design.
When you use real-world insights early, you design studies that are more practical, more patient-friendly, and more reflective of how therapies will actually perform.


Clinical trials are essential — but patients don’t live in controlled environments. They receive care in busy clinics, manage other conditions, and balance treatment with everyday life.
Real-World Data (RWD) helps bring that reality into trial design.
When you use real-world insights early, you design studies that are more practical, more patient-friendly, and more reflective of how therapies will actually perform.
Instead of guessing what your patient population looks like, RWD shows you:
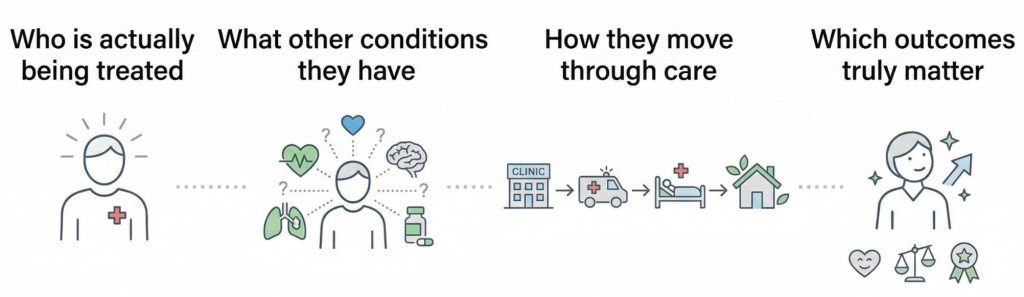
This leads to better eligibility criteria, more realistic enrollment goals, and fewer surprises after launch.
Many trials unintentionally make participation harder than it needs to be.
Real-World Data helps you:

Randomized controlled trials are powerful — but they’re often narrow by design.
Real-World Data expands the lens.
It shows how therapies perform across broader, more diverse populations — including patients who might not qualify for traditional trials. It captures long-term outcomes, safety trends, and effectiveness in routine care settings.
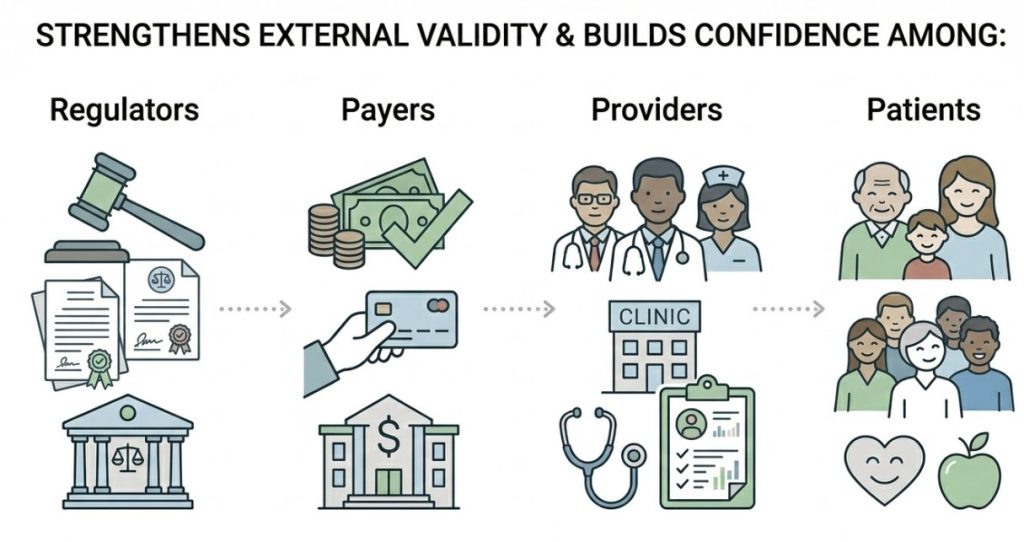
The question shifts from “Does it work under ideal conditions?” to “How does it perform in the real world?”
That distinction matters.
When RWD is integrated early in development, it can:
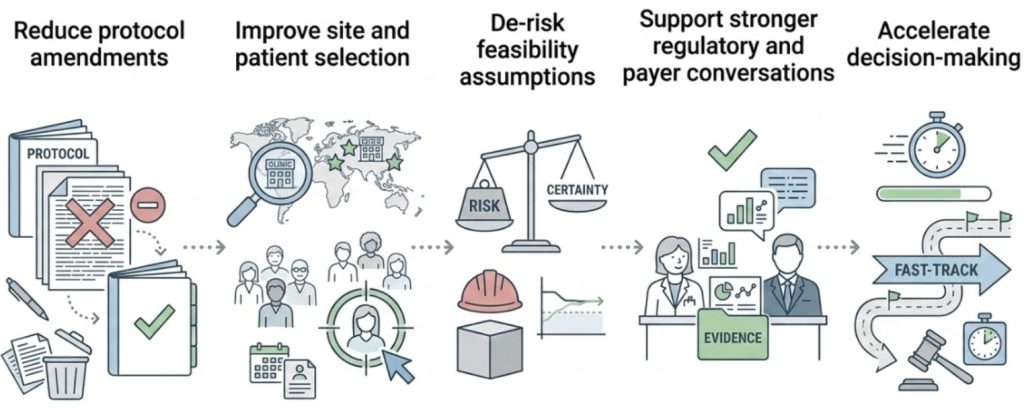
It becomes more than a post-approval evidence tool — it becomes a strategic design asset.
Your therapy won’t live inside a protocol. It will live in busy clinics, community practices, and patients’ daily lives.
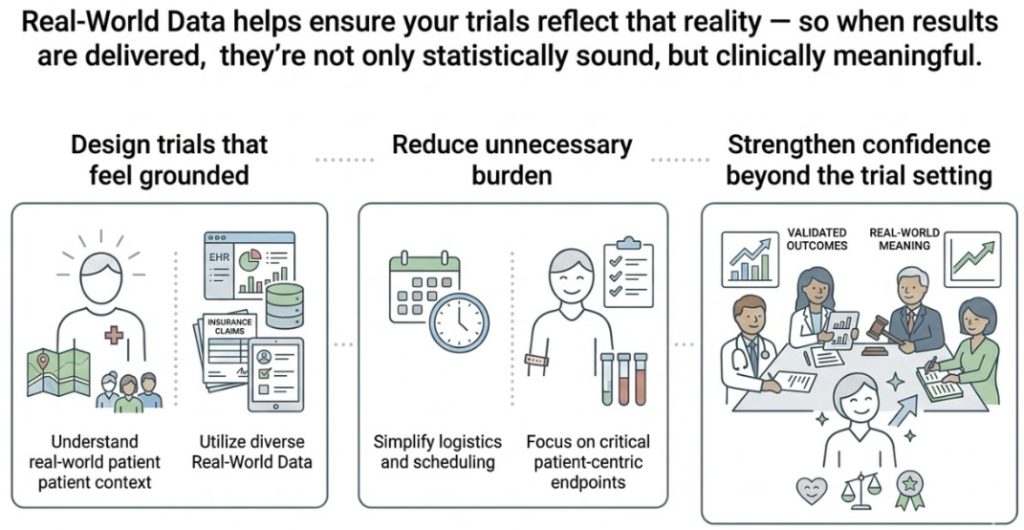
Because research works best when it reflects the real world.